How can stainless steel corrode?
All “stainless” steels can corrode under the wrong conditions. Remember that most of the metal is still iron and can corrode or rust depending on how much stress it is exposed to. One of the worst things you can do to stainless steel is expose it to chlorides. The better grades can withstand a lot of abuse, but most grades can eventually fail when exposed to chlorides in bad atmospheres. Some grades of “stainless” steel have very poor resistance to corrosion. They are marginally better than carbon steel, but when correctly hardened and passivated or electropolished can give fairly good performance. The grade of steel must reflect the exposure conditions and performance required.
 Corrosion Stainless Steel Pipe
Corrosion Stainless Steel Pipe
The main reason for the existence of the stainless steels is their resistance to corrosion. Chromium is the main alloying element, and the steel should contain at least 11 %. Chromium is a reactive element, but it and its alloys passivate and exhibit excellent resistance to many environments. Higher Cr contents may be necessary for to improve the corrosion resistance of stainless steels in more aggressive media. Among other alloying elements, Nickel is the most important : it is added to control the alloy microstructure and to improve the corrosion resistance in acidic or caustic media. The addition of other elements such as Molybdenum, Tungsten, Copper, Silicon, Titanium, Niobium, Nitrogen enables a wide range of properties to be obtained.
In metallurgy, stainless steel, also known as inox steel or inox from French “inoxydable”, is defined as a steel alloy with a minimum of 10.5 or 11% chromium content by mass.Stainless steel does not stain, corrode, or rust as easily as ordinary steel, but it is not stain-proof. It is also called corrosion-resistant steel or CRES when the alloy type and grade are not detailed, particularly in the aviation industry.
There are different grades and surface finishes of stainless steel to suit the environment the alloy must endure. Stainless steel is used where both the properties of steel and resistance to corrosion are required.
Stainless steel differs from carbon steel by the amount of chromium present. Unprotected carbon steel rusts readily when exposed to air and moisture. This iron oxide film (the rust) is active and accelerates corrosion by forming more iron oxide.
Stainless steels contain sufficient chromium to form a passive film of chromium oxide, which prevents further surface corrosion and blocks corrosion from spreading into the metal’s internal structure. Passivation only occurs if the proportion of chromium is high enough. All stainless steels contain some carbon.
Even though it is difficult to get much less than about 0.03 % and sometimes carbon is deliberately added up to 1.00% or more, modern melting processes allow to reduce the content below 0.015 or even 0.010%, providing stainless steels with superior resistance to intergranular corrosion.
The more carbon there is, the more chromium must be used, because carbon can take from the alloy about seventeen times its own weight of chromium to form carbides.
Chromium carbide is of little use for resisting corrosion. The carbon, of course, is added for the same purpose as in ordinary steels to make the alloy stronger.
Nitrogen is often added in modern stainless steels (especially in duplex grades or in highly alloyed austenitics); this element provides high mechanical properties, better resistance to pitting initiation and more generally improves the corrosion resistance thanks to a better microstructural stability. In 1913, English metallurgist Harry Brearly, working on a project to improve rifle barrels, accidentally discovered that adding chromium to low carbon steel gives it stain resistance. In addition to iron, carbon, and chromium, modern stainless steel may also contain other elements, such as nickel, niobium, molybdenum, and titanium. Nickel, molybdenum, niobium, and chromium enhance the corrosion resistance of stainless steel.
It is the addition of a minimum of 12% chromium to the steel that makes it resist rust, or stain ‘less’ than other types of steel. The chromium in the steel combines with oxygen in the atmosphere to form a thin, invisible layer of chrome-containing oxide, called the passive film.
The sizes of chromium atoms and their oxides are similar, so they pack neatly together on the surface of the metal, forming a stable layer only a few atoms thick. If the metal is cut or scratched and the passive film is disrupted, more oxide will quickly form and recover the exposed surface, protecting it from oxidative corrosion.
The passive film requires oxygen to self-repair, so stainless steels have poor corrosion resistance in low-oxygen and poor circulation environments. In seawater, chlorides from the salt will attack and destroy the passive film more quickly than it can be repaired in a low oxygen environment. From a microstructural point of view, alloying elements may be classified as either Austenite formers (Nickel, Nitrogen, Carbon) or Ferrite formers (Chromium and Molybdenum). Depending upon the ratio between austenite former elements and ferrite former elements, the resulting microstructure may be Ferritic (Body-Centered-Cubic crystal) or Austenitic (Face-Centered-Cubic crystal). The Martensitic structure (Close-Packed Hexagonal crystal) is the result of the transformation of austenite to martensite during cooling or during strong cold working. The Duplex microstructure is a mixture of Austenite and Ferrite (generally about 50/50%) which is the result of an accurate balance between Austenite and Ferrite former alloying elements.
Types of Stainless Corrosion
According to the DOD Technical Bulletin Corrosion Detection and Prevention there are 8 separate types of corrosion, with only a few having a major impact on stainless steel. Please be advised the descriptions below are extremely brief and written in laymen terms. Before acting on any particular application, qualified advice particular to such application should be obtained.
1. Uniform Attack - also known as general corrosion, this type of corrosion occurs when there is an overall breakdown of the passive film. The entire surface of the metal will show a uniform sponge like appearance. Halogens penetrate the passive film of stainless and allow corrosion to occur. These halogens are easily recognizable, because they end with “-ine”. Fluorine, chlorine, bromine, iodine and astatine are some of the most active.
2. Crevice Corrosion- this is a problem with stainless fasteners used in seawater applications, because of the low PH of salt water. Chlorides pit the passivated surface, where the low PH saltwater attacks the exposed metal. Lacking the oxygen to re-passivate, corrosion continues. As is signified by its name, this corrosion is most common in oxygen restricted crevices, such as under a bolt head.
Crevice corrosion is the second most common mode of failure of stainless steels, with pitting corrosion being the most common. Crevice corrosion occurs at locations where there is a small gap, or crevice, between the stainless steel article and another item. This second item can commonly be an insulator such as a gasket or a rubber separator although it could be metallic. The actual geometry of the small gap is critical for the onset of crevice corrosion. The gap has to be just big enough to allow an aggressive liquid to penetrate into the gap but not big enough to allow material to be flushed out of the gap due to flow or convection in the fluid. The mechanism of crevice corrosion is well-known, with the first stage being the accumulation of aggressive ions (such as chloride ions) in the gap and the exclusion of oxygen from the solution within the gap. This results in a fixed anode forming in the crevice with the material outside of the crevice becoming a fixed cathode. Corrosion occurs within the crevice for two reasons, firstly a passive film breaks down due to the exclusion of oxygen and secondly the corrosion reactions at the anodic site produce a change in the pH which means that the crevice becomes increasingly more acidic with time. Good design is one of the best ways to avoid crevice corrosion, with materials selection coming a close second. Crevice corrosion is most severe in acid conditions in chloride containing, non flowing or stagnant environments. Cathodic protection can reduce the levels of both pitting corrosion and crevice corrosion by making the anodic area more alkaline. Arranging for the environment to have greater flow will also cut down the effects of both forms of localised corrosion. Other passive materials such as aluminium and its alloys are susceptible to both crevice corrosion and pitting corrosion. Pitting and crevice corrosion of aluminium occurs by similar mechanisms to that of stainless steel.
3. Pitting- Stainless that had had its passivation penetrated in a small spot becomes an anodic, with the passivated part remaining a cathodic, causing a pit type corrosion.
The most usual type of localised corrosion attack on stainless steel which results in perforation of tanks, vessels and pipe walls is pitting corrosion. It takes the form of small diameter but deep holes. These are usually less than 1mm in diameter and can penetrate many millimeters into the metal.
The pits are the anodes in the corrosion reaction and the remainder of the surface is the cathode. Pits initiate at defects in the protective oxide layer, or passive layer, on the surface. Commonly, these defects are inclusions that form from impurities, such as sulphur, in the steel. These impurities can result in very local depletion of the alloying elements, thereby preventing the protective oxide from being totally uniform. The conditions that lead to pitting are moderately high temperatures, high concentrations of chloride ions (from salt), other halides such as fluorides, bromides and iodides. Acidic conditions (low pH) also aid the growth of the pits, which are themselves acidic. This acidity within the pit is the reason why pits can continue to grow once they have been initiated.
4. Galvanic Corrosion - Placing 2 dissimilar metals in a electrolyte produces an electrical current. A battery incorporates this simple philosophy in a controlled environment. The current flows from the anodic metal and towards the cathodic metal, and in the process slowly removes material from the anodic metal. Seawater makes a good electrolyte, and thus, galvanic corrosion is a common problem in this environment. 18-8 series stainless fasteners that work fine on fresh water boats, may experience accelerated galvanic corrosion in seawater boats, and thus it is suggested you examine 316 stainless.
5. Intergranular Corrosion- all austentic stainless steels contain a small amount of carbon. At extremely high temperature, such as welding, the carbon forces local chrome to form chromium carbide around it, thus starving adjacent areas of the chrome it needs for its own corrosion protection.
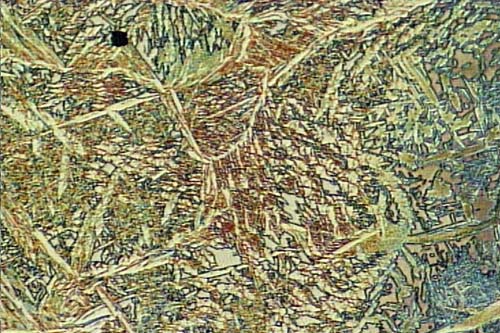
When welding, it is recommended you consider low carbon stainless such as 304L or 316L. Sensitization of stainless steel is a form of intergranular corrosion or grain boundary attack which results in complete grains of material falling out from the surface of the metal in the manner shown in the above photograph. When this happens in welded areas it is often referred to as weld decay. Where the sensitisation is related to a band across the component it is called Knife-line attack, as historically a heated band close to the handle of the stainless steel knife tended to lose hole grains fromm the surface which left a blackened pit. For 316 stainless steel sensitisation can occur when the steel is heated to a temperature between 480 deg C and about 900 deg C. At the higher temperatures sensitization can initiate after as little as three minutes but at the lower temperatures times of more than 1hour are required. Sensitisation results in corrosion as the grain boundaries become depleted in chromium due to the formation along the grain boundary of intermetallic carbides. Six carbon atoms take 23 chromium atoms out of the alloy.This can result in the chromium content being reduded from 18 % down to 12% locally. When sensitised stainless steels encounter a corrosive environment, the centre of the grain becomes a cathode & the grain boundary becomes a very active local anode site. The process can have a long initiation period as it can take quite a time for the surface grains to become undermined. However when the grains become loose they fall out of the surface as metallic flakes & leave a blacked pitted surface.
6. Selective Leaching - Fluids will remove metal during a de-ionization or de-mineralization process. This usually happens inside a pipe and is rarely a fastener problem.
7. Erosion Corrosion - This corrosion happens when the velocity of an abrasive fluid removes the passivation from a stainless. Again, this is almost exclusively limited to pipe interiors and rarely a fastener problem.
8. Stress Corrosion - Also called stress corrosion cracking or chloride stress corrosion. Chlorides are probably the single biggest enemy of stainless steel. Next to water, chloride is the most common chemical found in nature. In most environments, the PPM are so small the effects on stainless are minute. But in extreme environments, such as indoor swimming pools, the effects can be extreme and potentially dangerous. If a stainless part is under tensile stress, the pitting mentioned above will deepen, and cracking may take place. If you are using stainless steel bolts under tensile stress, in an environment where chlorine corrosion is likely, you should examine the potential for stress corrosion cracking carefully.
According to a NACE International & CC Technologies study, corrosion costs the United States $276,000,000,000 annually. That’s $276 billion and 4.2% of the nations GNP. In the power generation and transmission industry alone, it is estimated that nearly 8% of the typical electric bill is attributed to the cost of corrosion. It is unknown how many lives are lost annually due to corrosion but the number could be frighteningly high. Extreme examples thru the years include collapsed bridges and jet airliner crashes.
You might also like
| What is Stainless Steel? Stainless Steel - A Definition Stainless... | Austenite - Gamma Iron Austenite - a Definition Austenite also... | Microstructure of Metals Microstructure of Metals Microstructure... | Galvanized Steel - a Definition What is Galvanized Steel ? Sheet, strip,... |



 Alloy Suppliers
Alloy Suppliers
 Aluminum
Aluminum
 Aluminum Extrusions
Aluminum Extrusions
 Copper-Brass-Bronze
Copper-Brass-Bronze
 Nickel
Nickel
 Magnets
Magnets
 Stainless Steel
Stainless Steel
 Stainless Steel Tubing
Stainless Steel Tubing
 Steel Service Centers
Steel Service Centers
 Titanium
Titanium
 Tungsten
Tungsten
 Wire Rope
Wire Rope